Cellular energetic metabolism is highly dependent on mitochondrial function. Reactive oxygen species (ROS) are known to participate as signal messengers in diverse physiological networks and to be determinant in a variety of pathologies, such as diabetes, tumor progression, ischemic events, neurodegeneration and aging [1] .
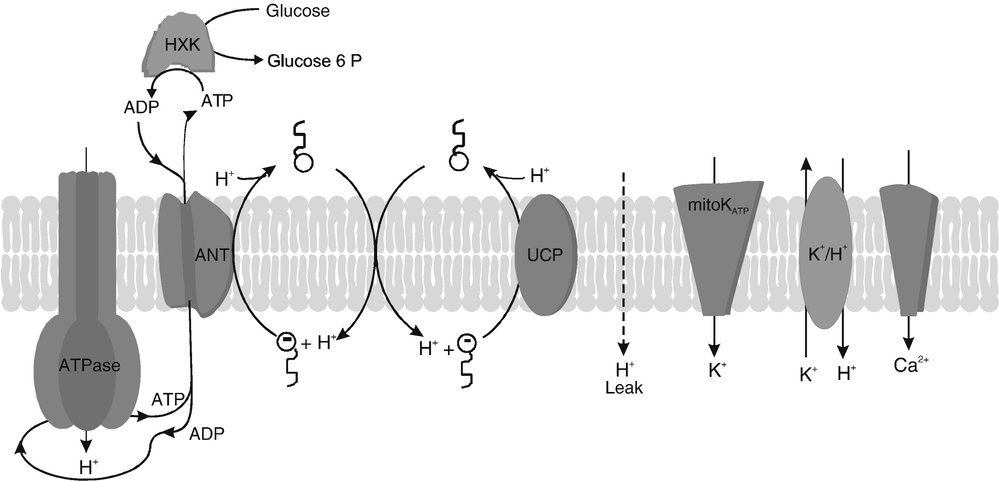
During mitochondrial oxidative phosphorylation can be uncoupled when respiratory complexes of the electron transport chain translocate protons to mitochondrial matrix. This proton leak means an energetic impairment due to heat production instead of completing ATP synthesis process [2].
Spatiotemporal equilibrium between ROS concentration and pH, modulated by proton leak, can affect organelle homeostasis as mitochondrial-associated membranes (MAMs) regulate cell metabolism. Thus, understanding how intracellular ROS and pH are regulated can offer insights in the signaling pathways connecting the energy-dependent endoplasmic reticulum (ER) function, lysosomal activity or lipid synthesis. Novel tools for are required to perform real-time dynamic assays where different intracellular analytes can be studied simultaneously to improve our understanding of metabolic processes. SPAchip® nanodevices can help due to their multiplexing possibilities and their lack of cytotoxicity.
[1]J. T. Konno, E. P. Melo, J. E. Chambers, and E. Avezov, “Intracellular sources of ROS/H2O2 in health and neurodegeneration: Spotlight on endoplasmic reticulum,” Cells, vol. 10, no. 2, pp. 1–26, 2021.nn[2] A. J. Kowaltowski, N. C. de Souza-Pinto, R. F. Castilho, and A. E. Vercesi, “Mitochondria and reactive oxygen species,” Free Radic. Biol. Med., vol. 47, no. 4, pp. 333–343, 2009.nn nnnn
nnnnnnn






